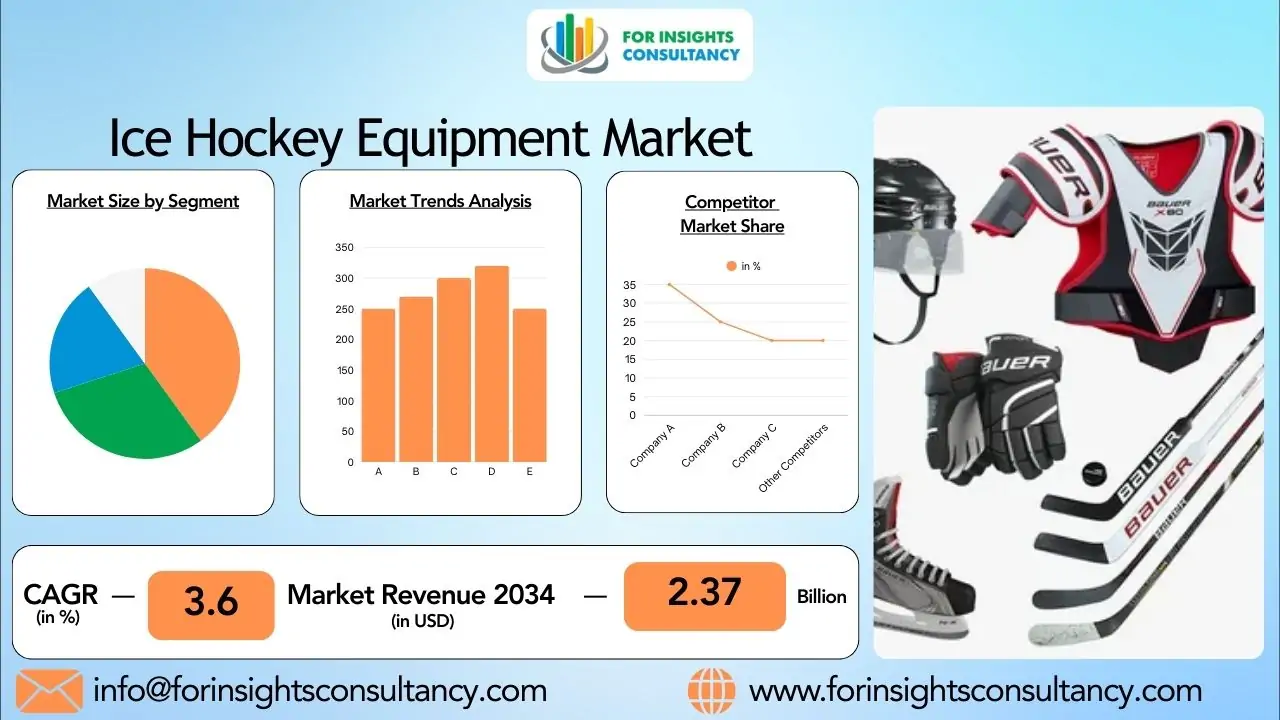
Afluria Market 2026: Rising Demand for Seasonal Influenza Vaccines
The global Afluria market — focused on seasonal influenza vaccination — is expected to grow strongly by 2026.
Introduction
The Afluria market is part of the broader seasonal influenza vaccine landscape and centers on the uptake of Afluria — an inactivated influenza vaccine used for active immunization against specific seasonal flu virus strains. Afluria is licensed for use in individuals six months of age and older and is typically administered annually to stimulate immune protection against circulating influenza viruses.
Market growth through 2026 is driven by recurring influenza outbreaks, public health vaccination policies, and expanding immunization programs across age groups including pediatric, adult, and geriatric populations.
Key Market Drivers
Increasing Prevalence of Seasonal Influenza
Seasonal influenza remains a major global health challenge, causing millions of illnesses and significant hospitalizations annually. The broad circulation of influenza A and B viruses underscores the need for effective preventive vaccines such as Afluria, which boost the body’s immune response against these infections.
Preventive Healthcare Awareness and Policy Support
Heightened public focus on preventive care, stronger public health vaccination campaigns, and improved health education are encouraging higher uptake of flu vaccines. Governments and healthcare organizations are emphasizing annual immunization schedules to reduce severe flu outcomes and healthcare burden.
Geriatric Demographic Growth
The global elderly population — more susceptible to severe influenza complications — continues to expand, increasing demand for annual influenza vaccinations like Afluria. Older adults often represent a key target demographic for seasonal flu immunization.
Expanded Vaccine Distribution Channels
Distribution through hospital pharmacies, clinics, retail pharmacies, and government immunization programs is enhancing access to Afluria vaccines in both developed and emerging markets. Improved cold chain logistics ensures better stability and reach across regions.
Types of Afluria Vaccine Options
The Afluria vaccine portfolio typically includes:
Quadrivalent Formulations
Designed to protect against four influenza virus strains — two influenza A strains and two influenza B strains — offering broader seasonal coverage.
Trivalent Formulations
Targeting three strains, including one influenza B strain in addition to the two influenza A strains. While less broad than quadrivalent vaccines, they remain widely used depending on regional immunization policies.
Vaccine Presentations
Afluria is available in pre-filled syringes and multi-dose vials, offering convenient options for mass immunization campaigns and routine clinical use.
Regional Market Insights
North America
North America remains the largest regional market for Afluria vaccines, supported by established vaccination schedules, robust public health programs, and strong healthcare infrastructure.
Asia-Pacific
The Asia-Pacific region is poised for the fastest growth during the forecast period due to expanding healthcare investments, increasing vaccine coverage targets, and growing awareness of influenza risks.
Europe and Other Regions
Europe also shows steady growth supported by supportive government immunization policies. Meanwhile, regions such as Latin America, the Middle East, and Africa are working to improve vaccine distribution systems and cold chain capacity.
Market Challenges
Despite strong prospects, the market faces several challenges:
- Tariff and supply chain pressures that may impact vaccine costs and procurement budgets in some regions.
- Seasonal variability of influenza virus strains, requiring annual reformulation and distribution planning.
- Vaccine hesitancy and coverage gaps in certain populations.
Efforts to address these, including education campaigns and distribution improvements, are key to sustaining market growth.
Emerging Trends in the Afluria Market
Several trends are shaping the future of the Afluria market:
Advancements in vaccine formulation technologies designed to improve immunogenicity and stability.
Expanded retail pharmacy vaccination programs to improve access and coverage.
Nano-sized particle vaccines and next-generation flu vaccine research that may influence competitive dynamics over the long term.
Afluria Market Outlook 2026
The Afluria market continues to grow in tandem with broader influenza vaccine demand globally. Rising flu infection rates, expanded immunization initiatives, and demographic trends are expected to support sustained demand through 2026. With ongoing investments in distribution infrastructure, preventive healthcare awareness, and product innovation, the Afluria market is poised for long-term expansion.