In Vitro Diagnostics Market 2026: Technology Trends and Industry Analysis
In Vitro Diagnostics Market Growth Analysis Research Report By Product/Service: Reagents & Kits, Instruments/Analyzers, Software & Services, By Technology: Immunoassays, Clinical Chemistry, Molecular Diagnostics, Hematology, Microbiology
Market Overview
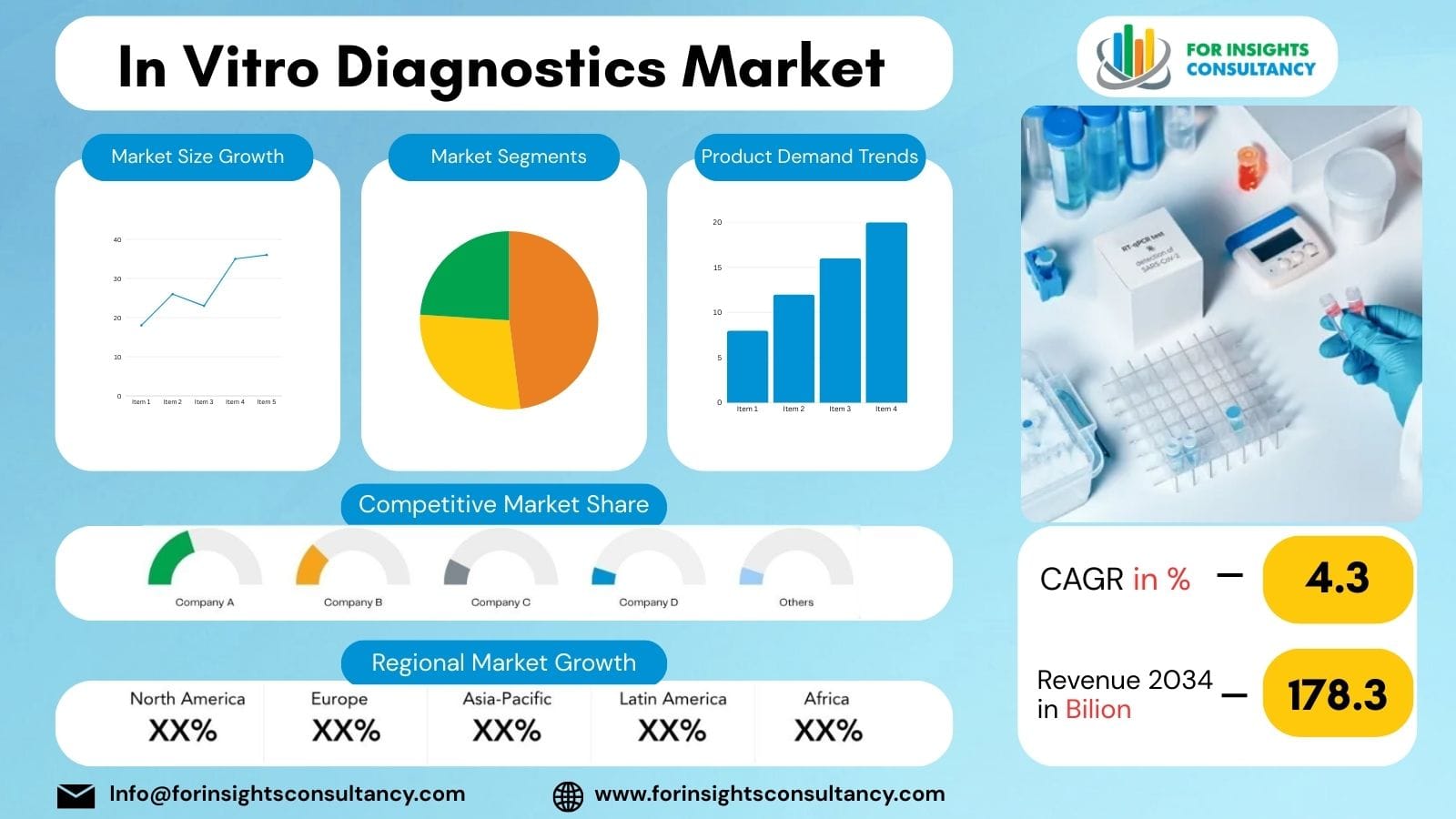
The In Vitro Diagnostics Market in 2026 is expected to witness strong expansion as healthcare systems increasingly rely on advanced diagnostic technologies for early disease detection and personalized treatment. In vitro diagnostics (IVD) involve medical tests performed on biological samples such as blood, urine, and tissues outside the human body to diagnose diseases and monitor health conditions.
The global In Vitro Diagnostics Market is projected to grow at a steady CAGR through 2030, driven by rising prevalence of chronic diseases, increasing demand for rapid diagnostic solutions, and advancements in molecular diagnostics and point-of-care testing technologies. Healthcare providers are adopting innovative diagnostic tools to improve clinical decision-making and patient outcomes.
Growing investments in healthcare infrastructure, increasing awareness of preventive healthcare, and continuous innovation in diagnostic technologies are expected to further accelerate the growth of the in vitro diagnostics industry.
Key Growth Drivers
Rising Prevalence of Chronic and Infectious Diseases
Chronic diseases such as diabetes, cancer, and cardiovascular disorders are increasing worldwide. In vitro diagnostic tests help in early disease detection, treatment monitoring, and disease management.
Growing Demand for Early Disease Detection
Healthcare systems are focusing on preventive healthcare and early diagnosis to reduce treatment costs and improve patient outcomes. This trend is driving the adoption of advanced diagnostic technologies.
Advancements in Molecular Diagnostics
Technological advancements in molecular diagnostics, genomics, and biomarker identification are improving the accuracy and speed of diagnostic testing.
Increasing Adoption of Point-of-Care Testing
Point-of-care diagnostic devices allow rapid testing at hospitals, clinics, and home settings. These technologies reduce diagnostic turnaround time and improve patient care.
Market Challenges
High Cost of Advanced Diagnostic Equipment
Sophisticated diagnostic instruments and molecular testing platforms can be expensive, which may limit adoption in low-resource healthcare systems.
Regulatory Compliance and Approval Processes
Diagnostic products must comply with strict regulatory standards and clinical validation requirements, which can delay product approvals and market entry.
Emerging Market Trends
Growth of Personalized Medicine
Personalized medicine is gaining popularity as healthcare providers use genetic and molecular data to tailor treatments for individual patients. In vitro diagnostics play a crucial role in enabling precision medicine.
Expansion of Digital Diagnostics
Digital health technologies, including artificial intelligence and cloud-based diagnostic platforms, are improving diagnostic accuracy and enabling remote healthcare services.
Increasing Use of Companion Diagnostics
Companion diagnostics are becoming essential in identifying patients who can benefit from specific therapies, especially in oncology treatments.
Regional Analysis
North America
North America holds a major share of the In Vitro Diagnostics Market due to advanced healthcare infrastructure, strong research capabilities, and high adoption of innovative diagnostic technologies.
Europe
Europe is experiencing steady market growth supported by government healthcare initiatives, advanced medical research, and increasing demand for diagnostic testing.
Asia-Pacific
Asia-Pacific is expected to witness the fastest growth in the in vitro diagnostics market due to rising healthcare investments, growing population, and increasing awareness of disease prevention in countries such as China and India.
Latin America and Middle East
These regions are gradually expanding their healthcare infrastructure and diagnostic capabilities, contributing to the growth of the in vitro diagnostics industry.
Competitive Landscape
The in vitro diagnostics industry is highly competitive, with global healthcare companies investing in research and development to introduce innovative diagnostic solutions.
Key companies operating in the market include:
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- Danaher Corporation
- Thermo Fisher Scientific
- bioMérieux
These companies focus on developing advanced diagnostic technologies, molecular testing platforms, and digital health solutions to strengthen their global market presence.
In Vitro Diagnostics Market Size in 2026
The In Vitro Diagnostics Market in 2026 is expected to grow significantly as healthcare providers adopt advanced diagnostic technologies for early disease detection and improved patient care. Innovations in molecular diagnostics, point-of-care testing, and digital healthcare are supporting industry expansion.
Future Market Outlook
The future of the In Vitro Diagnostics Market appears highly promising as diagnostic technologies continue to evolve. Growing investments in healthcare research, increasing demand for precision medicine, and advancements in artificial intelligence are expected to accelerate innovation in the diagnostics sector.
As healthcare systems shift toward preventive and personalized care models, in vitro diagnostics will remain a critical component of modern medical practice.
Frequently Asked Questions (FAQs)
What is the In Vitro Diagnostics Market size in 2026?
The In Vitro Diagnostics Market in 2026 is expected to grow steadily due to increasing demand for advanced diagnostic technologies and rising prevalence of chronic diseases.
What are the key drivers of the In Vitro Diagnostics Market?
Key drivers include rising chronic disease prevalence, advancements in molecular diagnostics, increasing demand for early disease detection, and growing adoption of point-of-care testing.
Which region dominates the In Vitro Diagnostics Market?
North America dominates the market due to its advanced healthcare infrastructure, strong research capabilities, and early adoption of innovative diagnostic technologies.
What are common types of in vitro diagnostic tests?
Common IVD tests include molecular diagnostics, immunoassays, clinical chemistry tests, hematology tests, and point-of-care diagnostic tests.
What are the future trends in the In Vitro Diagnostics Market?
Future trends include digital diagnostics, AI-powered healthcare technologies, personalized medicine, and increasing adoption of companion diagnostics.